By Our Correspondent
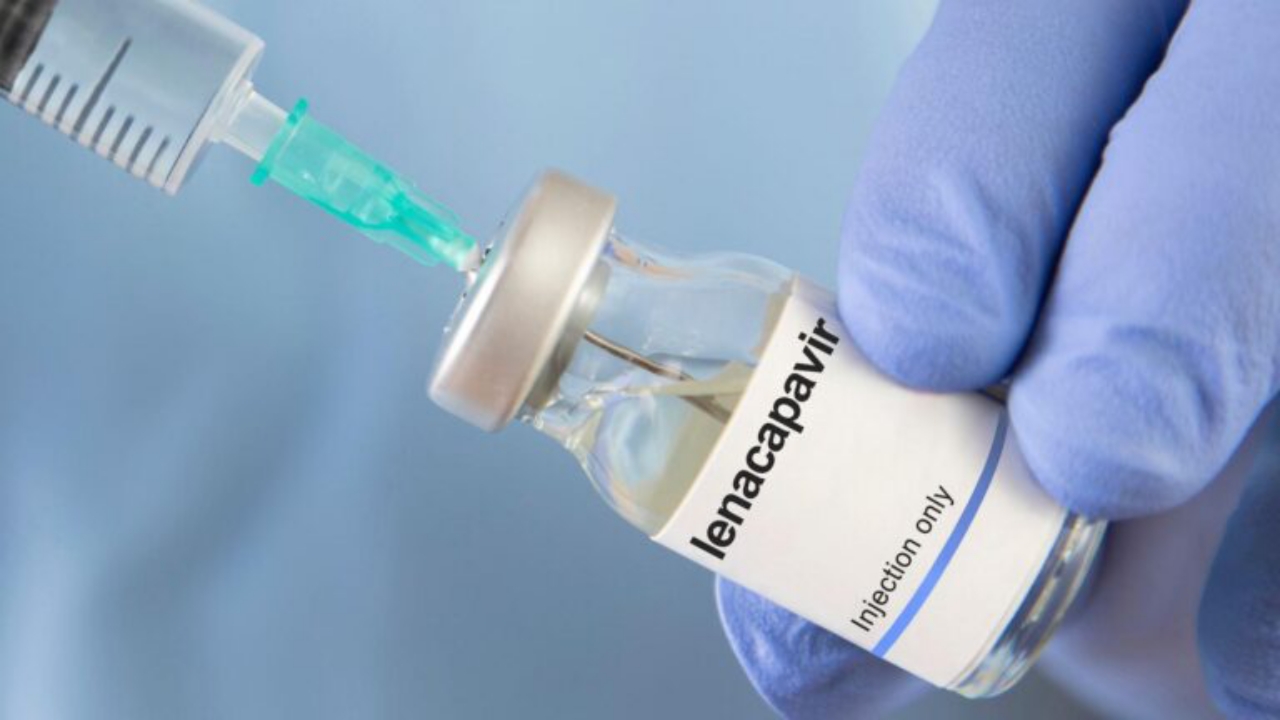
National News – Demand is rapidly increasing across Nigeria for Lenacapavir, a newly introduced injectable drug designed to prevent infection from Human Immunodeficiency Virus (HIV).
Many Nigerians, especially individuals familiar with pre-exposure prophylaxis (PrEP), have begun approaching health facilities and non-governmental organisations in anticipation of the drug’s official rollout.
Lenacapavir is a long-acting HIV prevention injection administered twice a year, offering an alternative to daily PrEP pills.
The medication, developed by Gilead Sciences, was endorsed by the World Health Organization (WHO) in 2025 as part of global efforts to strengthen HIV prevention strategies.
Health experts say the injection works by preventing the virus from establishing infection in the bloodstream after exposure.
According to recent estimates from Nigeria’s National Agency for the Control of AIDS (NACA) and the Joint United Nations Programme on HIV/AIDS (UNAIDS), about two million Nigerians are currently living with HIV/AIDS.
In response, the Federal Government has begun preparations to introduce the injection in selected pilot states.
Officials confirmed that Nigeria expects about 52,000 doses, with 11,520 already delivered.
The initial rollout will take place in Lagos, Akwa Ibom, Anambra, Benue, Cross River, Ebonyi, Gombe, Kano, Kwara, and the Federal Capital Territory (FCT).
The drug will be administered free of charge at designated health facilities, including general hospitals.
Reports from several states indicate growing interest in the preventive injection.
Health workers, individuals in discordant relationships—where one partner is HIV-positive and the other negative—and other high-risk groups are among those seeking early access.
Some medical professionals have also expressed interest in receiving the injection as an added layer of protection.
However, health authorities stress that Lenacapavir is not a cure for HIV.
It is strictly designed for prevention among individuals who are not infected.
Those already living with HIV must continue lifelong antiretroviral therapy (ART).
Medical experts also noted that the injection is currently not recommended for pregnant women, and only trained healthcare personnel will administer it at approved centres.
Common side effects reported so far include mild swelling or pain at the injection site.
Pharmaceutical stakeholders have urged regulators to ensure that the drug’s nationwide deployment follows strict safety and approval processes to guarantee quality, effectiveness, and proper monitoring.